Background: Spontaneous intracranial hypotension (SIH) is a relatively rare but known pathology that results in brain herniation or sagging due to CSF hypovolemia. SIH can commonly be misdiagnosed when occurring with other pathologies including chronic subdural hematoma (CSDH). Neurological decline caused by SIH is rare, but is often rapid and much more severe.
Case description: A 69-year-old man was delivered to the emergency department for loss of consciousness and mild left hemiparesis. Brain computed tomography (CT) revealed increased density over basal cisterns suggestive of subarachnoid hemorrhage (SAH), and CSDH. Cerebral angiography showed dural arteriovenous fistula, but there was no apparent cerebral aneurysm. We first performed trans-arterial embolization using n-butyl cyanoacrylate, followed by bilateral hematoma evacuation. A postoperative CT showed that CSDH was evacuated, but the subdural space was filled with free air. A subsequent CT examination revealed mild re-accumulation of CSDH without significant brain compression. On postoperative day 14 the patient became progressively lethargic, for which he was transferred to the intensive care unit. As gadolinium-enhanced magnetic resonance imaging and radionuclide cisternography suggested the leakage of cerebrospinal fluid, an epidural catheter was inserted in the head- down positioned patient and an epidural infusion of saline was given for 3 days. The patient stabilized and regained consciousness over 2 weeks. The patient became neurologically intact with imaging showing almost complete parenchymal expansion and resolution of subdural fluid collection.
Conclusion: This is an instructive case because the patient deteriorated after the surgery for CSDH, but it is also a unique case because the deterioration occurred over 14 days. This report emphasizes the possibility that SIH should be taken into consideration when there is an unknown reason for early CSDH recurrence and neurological deterioration.
Keywords: Spontaneous intracranial hypotension, Continuous epidural saline infusion, Chronic subdural hematoma, Impaired consciousness
Abbreviations: SIH: Spontaneous Intracranial Hypotension, CSF: Cerebrospinal Fluid, CSDH: Chronic Subdural Hematoma, CT: Computed Tomography, MRI: Magnetic Resonance Imaging, SAH: Subarachnoid Hemorrhage,AVF: Arteriovenous Fistula
Spontaneous intracranial hypotension (SIH) is the result of abnormally low intracranial pressure and most commonly manifests as postural headaches.1 SIH is a relatively rare but known pathology that results in brain herniation or sagging due to cerebrospinal fluid (CSF) hypovolemia.2-5 However, SIH can commonly be misdiagnosed, when occurring with other pathologies attributed to Chiari malformations, compression from subdural hematomas, or possible malignancies leading to leptomeningeal enhancement.6 Neurological decline caused by SIH is rarely reported, but is often rapid and much more severe. Several reports have noted that lumbar drainage placed to assist brain relaxation during treatment of aneurysms causes unexplained neurological decline with imaging evidence of brain herniation.2,7-9 In this report, our case showed relatively delayed onset of the exacerbation of consciousness after a bilateral hematoma evacuation, and improvement of neurological status following continuous infusion of saline into the epidural space.
A 69-year-old man without apparent event of head trauma was presented to the department of otolaryngology for a 2-year history of tinnitus. Magnetic resonance imaging (MRI) showed bilateral fluid collection suspected of chronic subdural hematoma (CSDH). The patient was then referred to our department for further examination and treatment at 2 weeks prior to hospitalization. Because there was no neurological abnormality, we decided to have regular follow-ups. However, he was delivered to the emergency department for loss of consciousness on the day of admission.
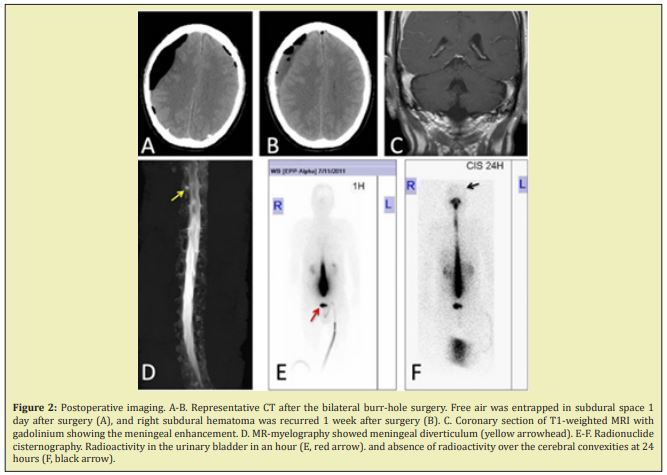
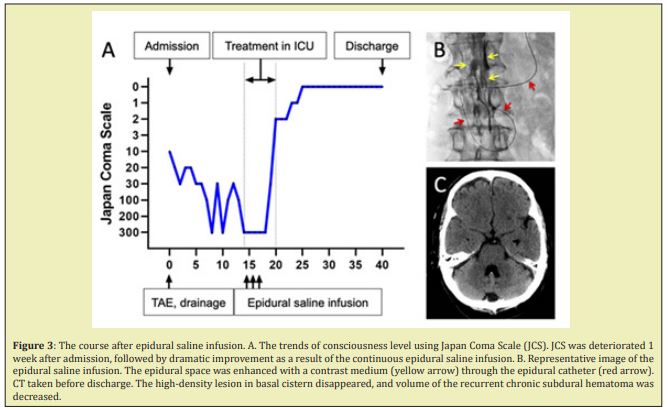
His Japan Coma Scale on admission was 10, and he had mild left hemiparesis. Brain computed tomography (CT) revealed increased attenuation over basal cisterns that was suggestive of subarachnoid hemorrhage (SAH) (Figure 1A), though there was no remarkable change in already known CSDH (Figure 1B). Cerebral angiography failed to demonstrate any apparent aneurysm or arteriovenous malformation, except for dural arteriovenous fistula (dural AVF) detected by super-selective left external carotid angiography (Figure 1C). The dural AVF existed between the Petro squamosal branch of left middle meningeal artery and the transverse-sigmoid sinus without retrograde flow in the venous sinus or retrograde cortical venous drainage. We first performed trans-arterial embolization using n-butyl cyanoacrylate at the concentration of 20%, achieving disappearance of the arteriovenous shunt (Figure 1D). Afterward, he received bilateral burr-hole surgery with placement of bilateral subdural drains. Postoperative CT showed evacuation of subdural fluid with a large amount of pneumocephalus, without significant brain expansion (Figure 2A). The patient was kept in a recumbent supine position because mild lethargy and confusion remained after the procedure, demonstrating a waxing and waning mental status. Subsequent imaging revealed mild re-accumulation of subdural fluid without significant brain compression (Figure 2B). He received several imaging examinations to detect the cause of deteriorated consciousness. Brain MRI with gadolinium-enhancement showed mild meningeal enhancement (Figure 2C). MRI myelography showed a small meningeal diverticulum (Figure 2D). Radionuclide cisternography showed early accumulation of nucleotide in the bladder (Figure 2E), and delayed migration of radioactivity over the cerebral convexities even at 24 hours post-injection (Figure 2F). On postoperative day 14, the patient became progressively lethargic, for which he had to be transferred to the intensive care unit for cardiorespiratory management (Figure 3A). Therefore, an epidural catheter was inserted in the head- down positioned patient and an epidural infusion of saline with an infusion rate of 20 ml/h was started immediately (Figure 3B). Over the next 3 days, the epidural infusion was maintained at this rate. The patient stabilized and regained consciousness over 2 weeks. From day 26, he could start his rehabilitation program without positional headache. Within a course of 6 weeks, the patient became neurologically intact, and the follow-up CT showed the disappearance of the high-density area in basal cistern and resolution of subdural fluid collections (Figure 3E). He was discharged from the hospital on day 40.

One of the challenges in our case was that the initial CT showed an “SAH-like” finding other than bilateral CSDH. CT findings of increased density in the basal cistern usually suggest SAH. However, diffuse cerebral edema, purulent meningitis, cerebral infarction, subdural hematoma, leakage of intravenous contrast into the subarachnoid space, status epilepticus, intrathecally administered contrast medium, polycythemia, and SIH were reported to mimic SAH.10 Ferrate, et al. suggested that patients with pseudo SAH should undergo conventional angiography in order to rule out an aneurysmal source of bleeding.11 As there is no doubt that SAH due to cerebral aneurysm or arteriovenous malformation is a critical disease, cerebral angiography was necessary to evaluate the cerebral arterial lesion. After excluding these vascular lesions, the “SAH-like” finding could be judged as “pseudo SAH” caused by compression of the cerebrum. Also, it seemed to be reasonable that the cause of loss of consciousness was CSDH because the CSDH case with the onset of unconscious attacks had been previously reported.12 Although SIH should have been one of the differential diagnoses, it was difficult to suspect at the time of admission.
According to the classification for dural AVF, the risk of intracranial hemorrhage from Borden type I (Cognard types I, IIa) lesions is extremely low in our case.13 A few reports suggested that iatrogenic dural AVF could be associated with surgical treatment for subdural hematomas.14,15 There was significant concern that shunt flow through the fistula might increase after surgery, which could cause brain edema or intracerebral hemorrhage. As it was difficult to predict the angiographical change of dural AVF after hematoma evacuation, it seemed to be better to conduct the trans-arterial obliteration with n-butyl cyanoacrylate for dural AVF prior to the burr-hole surgery for CSDH.
In our case, postoperative CT showed the bilateral pneumocephalus, implying the possibility of SIH. Sometimes free air gets into the subdural space after CSDH surgery, which is known as a predictive factor of recurrent CSDH.16 However, recent case reports have shown that the expanded pneumocephalus suggests the existence of SIH.17 Also, Takahashi, et al. suggested that physicians should pay attention to underlying SIH to avoid multiple surgery among the patients with refractory CSDH, especially in young to middle aged patients without preceding trauma or hematological disorders.18 Therefore, we started the conservative treatment for SIH, though we had not diagnosed his condition yet.
Supportive measures including bed rest and a generous oral fluid intake could improve symptoms caused by SIH over a period of several weeks.19,20 However, more invasive treatments are offered to those failing conservative treatments. The most commonly employed treatment and mainstay of therapy for SIH is by far the epidural blood patch.21 However, a retrospective study described that the lack of the site of leakage decreased the success rate of epidural blood patches.22 Other estimated weak points of epidural blood patches are the low number of hospitals where epidural blood patches can be performed, and the potential risk of adhesion in the epidural space. There are few case reports on the use of epidural saline infusion in SIH patients.23-25 In emergency situations, as in our case, epidural saline infusion can be used to rapidly stabilize the patient.19 The epidural saline infusion is technically safe because the placement of the epidural catheter is routinely applied by anesthesiologists. Because our case needed to stay in the intensive care unit, and because there was not an apparent leakage point of cerebrospinal fluid, the epidural saline infusion seemed to be an appropriate treatment option.
Most important to note from this case is that the possibility of SIH should be taken into consideration, especially when a postoperative CT shows pneumocephalus and neurological findings do not improve after the surgery for CSDH. When there is not an apparent leakage point of cerebrospinal fluid, the epidural saline infusion may be an appropriate treatment option.
We thank Herminio J Cardona (Northwestern University) for editing a draft of this manuscript.
None.
Author declares that there are no conflicts of interest.
- 1. Schievink WI. Spontaneous spinal cerebrospinal fluid leaks: a review. Neurosurg Focus. 2000;9(1):e8.
- 2. Bloch J, Regli L. Brain stem and cerebellar dysfunction after lumbar spinal fluid drainage: case report. J Neurol Neurosurg Psychiatry. 2003;74(7):992‒994.
- 3. Ferrante E, Savino A, Sances G, et al. Spontaneous intracranial hypotension syndrome: report of twelve cases. Headache. 2004;44(6):615‒622.
- 4. Miyazawa K, Shiga Y, Hasegawa T, et al. CSF hypovolemia vs intracranial hypotension in "spontaneous intracranial hypotension syndrome". Neurology. 2003;60(6):941‒947.
- 5. Park ES, Kim E. Spontaneous intracranial hypotension: clinical presentation, imaging features and treatment. J Korean Neurosurg Soc. 2009;45(1):1‒4.
- 6. Schievink WI. Misdiagnosis of spontaneous intracranial hypotension. Arch Neurol. 2003;60(12):1713‒1718.
- 7. Hirono S, Kawauchi D, Higuchi Y, et al. Life-Threatening Intracranial Hypotension after Skull Base Surgery with Lumbar Drainage. J Neurol Surg Rep. 2015;76(1):e83‒e86.
- 8. Kim YS, Kim SH, Jung SH, et al. Brain stem herniation secondary to cerebrospinal fluid drainage in ruptured aneurysm surgery: a case report. Springerplus. 2016;5:247.
- 9. Samadani U, Huang JH, Baranov D, et al. Intracranial hypotension after intraoperative lumbar cerebrospinal fluid drainage. Neurosurgery. 2003;52(1):148‒151.
- 10. Lin CY, Lai PH, Fu JH, et al. Pseudo-subarachnoid hemorrhage: a potential imaging pitfall. Can Assoc Radiol J. 2014;65(3):225‒231.
- 11. Ferrante E, Regna Gladin C, Arpino I, et al. Pseudo-subarachnoid hemorrhage: a potential imaging pitfall associated with spontaneous intracranial hypotension. Clin Neurol Neurosurg. 2013;115(11):2324‒2328.
- 12. Yoshida R, Otomo E, Kusunoki S. A case of chronic subdural hematoma with frequent unconscious attacks. Rinsho Shinkeigaku. 1986;26(1):35‒38.
- 13. Zipfel GJ, Shah MN, Refai D, et al. Cranial dural arteriovenous fistulas: modification of angiographic classification scales based on new natural history data. Neurosurg Focus. 2009;26(5):E14.
- 14. Mewada T, Ohshima T, Yamamoto T, et al. Usefulness of Embolization for Iatrogenic Dural Arteriovenous Fistula Associated with Recurrent Chronic Subdural Hematoma: A Case Report and Literature Review. World Neurosurg. 2016;92:584.e587-584.e510.
- 15. Pappas CT, Zabramski JM, Shetter AG. Iatrogenic arteriovenous fistula presenting as a recurrent subdural hematoma. Case report. J Neurosurg. 1992;76(1):134‒136.
- 16. Nakaguchi H, Tanishima T, Yoshimasu N. Relationship between drainage catheter location and postoperative recurrence of chronic subdural hematoma after burr-hole irrigation and closed-system drainage. J Neurosurg. 2000;93(5):791‒795.
- 17. Shin HS, Lee SH, Ko HC, et al. Extended Pneumocephalus after Drainage of Chronic Subdural Hematoma Associated with Intracranial Hypotension: Case Report with Pathophysiologic Consideration. J Korean Neurosurg Soc. 2016;59(1):69‒74.
- 18. Takahashi T, Senbokuya N, Horikoshi T, et al. Refractory chronic subdural hematoma due to spontaneous intracranial hypotension. No Shinkei Geka. 2007;35(8):799‒806.
- 19. Schievink WI, Meyer FB, Atkinson JL, et al. Spontaneous spinal cerebrospinal fluid leaks and intracranial hypotension. J Neurosurg. 1996;84(4):598‒605.
- 20. Srimanee D, Pasutharnchat N, Phanthumchinda K. Bilateral subdural hematomas and hearing disturbances caused by spontaneous intracranial hypotension. J Med Assoc Thai. 2009;92(11):1538‒1543.
- 21. D'Antona L, Jaime Merchan MA, Vassiliou A, et al. Clinical Presentation, Investigation Findings, and Treatment Outcomes of Spontaneous Intracranial Hypotension Syndrome: A Systematic Review and Meta-analysis. JAMA Neurol. 2021;78(3):329‒337.
- 22. Sencakova D, Mokri B, McClelland RL. The efficacy of epidural blood patch in spontaneous CSF leaks. Neurology 2001;57(10):1921‒1923.
- 23. Katayama T, Hirai S, Hamanaka Y, et al. Effect of the continuous epidural saline infusion for patients with postdural puncture headache after pulmonary resection. Kyobu Geka. 2011;64(12):1071‒1075.
- 24. Kawasaki S, Yamamoto Y, Sunami N, et al. Treatment of spontaneous intracranial hypotension with continuous epidural saline infusion: a case report. No To Shinkei. 1999;51(8):711‒715.
- 25. Rouaud T, Lallement F, Choui R, et al. Treatment of spontaneous intracranial hypotension by epidural saline infusion. Rev Neurol (Paris) 2009;165(2):201‒205.

