Introduction: ongenitalheartdefectsrepresentoneofthemainmalformationsdiagnosed prenatally. These are a major cause of neuro developmental disorders and it has even been proposed that structural brain alteration is found from early stages of fetal development.
Objective: The objective of this study was to review the existing evidence of prenatal diagnosis of structural braina lterations found in fetal brain magnetic resonance imaging and neurosonography in fetus with isolated congenital heart defect.
Materials and Methods: A search of Pubmed, Science Direct, Clinical Key and EBSCO databases was conducted in order to review study cases of fetuses with congenital heart defect between 18 and 37 week with Doppler ultrasonography, neurosonography and /or fetal brain magnetic resonance imaging.
Results: A total of 16 articles were selected and it was found that the fetuses with congenital heart defect present structural neurological alterations from intrauterine life showed by neurosonography and fetal brain magnetic resonance imaging.
Conclusion: The evidence emphasizes on the importance of studying in detail the central nervous system of fetuses with congenital heart defect and the relationship between structural brain alterations and neurodevelopmental disorders impairment since prenatal stages.
Keywords: Congenital heart defect, Fetal magnetic resonance, Neurosonography, Structural neurological alterations, Neurodevelopment.
Congenital heart defects(CHD) represent one of the mainmal formations diagnosed prenatally, affecting upto1% of pregnancies.1,2 These are a major cause of neuro developmental disorders (ND) that can be evidenced in the early years of childhood.3 ND alteration soccur in approximately 50% of live newborns with moderate to severe CHD diagnosed prenatally.4-7 However, the specific prenatal causes and the mechanisms of insult are largely unknown and it isnotyetclearifthestructuralbrainalterationsinfetuseswithisolated CHD evidenced in Doppler ultrasonography and magnetic resonance imaging (MRI) of the brain are directly related to ND alterations.6,8 In fetal brain MRI, brain abnormalities appear with a prevalence of 39%.9 Brain structural changes and ND alteration occur in different stages in both prenatal and postnatal l life and have been demonstrated by several studies.10 However, the number of studies that demonstrate the previous argument sin sufficient.3,5 There search focused on the alterations that occurred the corrective surgery and cardiopulmonary bypass with a perioperative perspective. Nevertheless, it was found that more than half of the children had cognitive and structural deficiencies prior to the surgical procedure, which is why it is raised with more evidence that central nervous system alterations occur since intrauterine life.9,11,12 It has been proposed that intrauterine structural brain alteration in fetuses with CHD occur prior to ND alteration.13,14 Frequent findings are white matter alterations, cortical maturation, strokes, hemorrhages and functional alterations before cardiac surgery.12,15 Considering that the cognitive development disorders have a prevalence of around 1% in developed countries and 2% in developing countries, it is important the diagnosis of structural and functional alterations of the brain parenchyma in the prenatal stage. The latter would facilitate a proper monitoring and timely intervention. Consequently, this will allow providing adequate counseling to families and achieving early interventions within the first years of life, which are crucial for ND. The objective of this systematic review was to analyze the existing evidence regarding the prenatal diagnosis of structural brain alterations evidenced in MRI and neurosonography (NS) in fetuses with congenital heart defect.
Design
Systematic literature search
Types of study to include
All epidemiological study designs were included with the exception the series and case reports.
Population
Fetuses with ultrasound diagnosis of CHD between 18 and 37 weeks of gestation without chromosomic alterations or associated extra cardiac malformations and with Doppler ultrasonography, NS and fetal brain magnetic resonance imaging.
Types of diagnostic tests
Fetal brain magnetic resonance imaging, ultrasound, feto placental Doppler
Search methods to identify the studies: Electronic searches: We identify the studies that meet the in clusion criteria in English or Spanish. We use a set of controlled and uncontrolled terms for “Fetus”, “Fetal”, “Heart Defects”, “Congenital”, “Magnetic Resonance Imaging”, “Neurological structure”, “Nervous system structure” and “Neuro developmental Disorders”, with field labels (title and abstract), proximity operators and Boolean operators. The search strategies are found in Annex1.
Specifically we use the following data bases:
• The Cochrane Central Register of Controlled Trials
• (CENTRAL, Ovid platform): inception to present.
• MEDLINE®, Ovid platform: inception to present.
• MEDLINE® In-Process & Other Non-Indexed Citations,
• Ovid platform: inception to present.
• MEDLINE® Daily Update, Ovid platform: inception to present.
• EMBASE, embase. Com platform: inception to present.
• LILACS, IAH xinterface: inception to present.
Data collection and analysis
Selection of studies
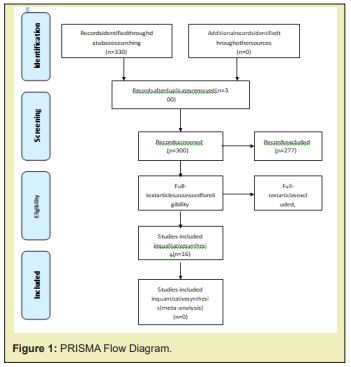
Two authors considered the inclusion and exclusion criteria of all the titles and abstracts found in the search strategy and carried out a qualitative analysis of them. All the potentially relevant studies were evaluated in full text. The entire selection process was documented in a PRISMA flow diagram.
Data extraction and management
An extraction format was designed and an author will collect the following information of each reference:
• Study design
• Year of publication
• Participants: characteristics
• Number of participants in each group
• Losses in the monitoring
• Tests used
• Definition and frequency of outcomes in each group
Risk of bias assessment
The authors independently assessed the risk of bias included according their designs. For randomized studies we will use the risk of bias assessment of Cochrane Collaboration. For observational studies we will use the Joanna Briggs Institute Check list.
Search results
Having into account the research question, a literature search was conducted finding 330 articles, of which 30 articles were ruled out for duplication, 277 for not answering their search question when reviewing the title and the abstracts, 1 for being a case report and 6 for being systematic reviews, leaving a total of 16 studies for the review (n=912cases) Figure 1:
• 6 case studies and controls (n=214cases)
• 9 cohort studies (n=565 cases)
• 1clinic altrial (n=133cases)
Studies out comes
For the qualitative analysis of the studies that met the inclusion criteria (16 studies), they were grouped by study type.
Case-control studies (n=6)
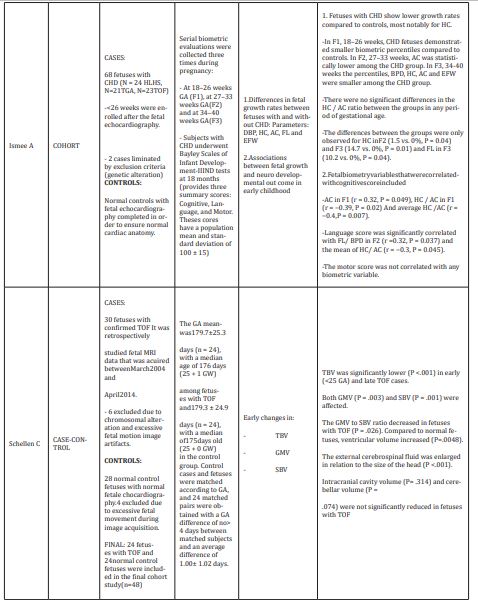
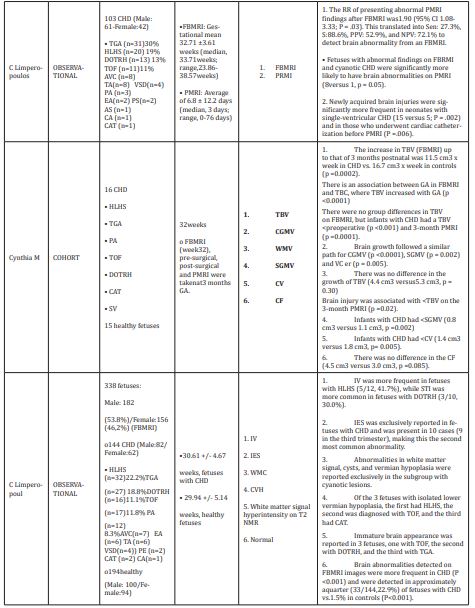
The case-control studies had in common the search for the association between alterations in brain growth (structural and functional) and CHD. It is increasingly recognized that the brain is abnormal in many newborns with CHD, which was previously attributed exclusively to postnatal cardiac surgery.9,11,12 Several theories have been proposed to explain the correlation between CHD and fetal ND alterations; however, it is not yet clear if these neurological structural alterations are found in all fetuses with CHD, in addition, if these are directly related to the ND alterations.6,8,16-18 Studies have found neurological alterations in utero with the use of fetal brain magnetic resonance imaging. Weissmann-Brenner A19 in their case-control study they report significantly higher rates of general cerebral, ventricular and cortex pathologies and altered biometric parameters in the cyanotic CHD group compared to the control group.19 The results suggest a pathogenesis in the uterus of the neurological and cognitive abnormalities found during childhood development. In the evaluation of neurological development, cerebral cortical development and regional growth of brain tissue have been studied as early and sensitive markers of brain growth failure in this and other high-risk populations. Similarly, Clouchoux Cetal,13 found that the volumes of white and gray matter were significantly lower in the group of Hypoplastic Left Heart Syndrome(HLHS) vs controls (p value<0.001), with a difference that became progressively higher after 30 weeks of gestation (GA).13 This study reports for the first time in vivo cortical development in fetuses with HLHS, finding that the cingulate sulcus was visible in controls from 25 GA, but not until 28 GA in fetuses with HLHS; the superior frontal sulcus was visible from 27 GA in control fetuses, but not until 30 GA in HLHS fetuses; likewise, the anterior ascending limb was visible from week 32 to 33 GA in controls, but only until 36GA in fetuses with HLHS. However, in this study, only fetuses with HLHS were evaluated, which prevents extrapolating the results too ther types of heart diseases. Previously, Limperopoulos C6 reported brain growth and metabolism between normal fetuses and those with CHD, with the use of three-dimensional volumetric magnetic resonance imaging (MRI) and proton magnetic resonance spectroscopy (H-MRS).6 This study found that brain volume and metabolism are abnormal in third trimester fetuses with some forms of CHD, particularly those with HLHS, transposition of the great arteries (TGA), and reduced flow through the systemic ventricle or antegrade around the aortic arch. In addition, quantitative magnetic resonance imaging provided deeper information about the timing of abnormalities that could disrupt normal fetal brain development. This study found that control fetuses had linear relationships between total brain volume (TBV) and GA (p value<0.001). Similarly, linear relationships between intracranial cavity volume (ICV) and GA (p value<0.001) with progressive increases in TBV and ICV during the third trimester. TBV and ICV were progressively smaller in fetuses with CHD relative to controls (p value<0.001). This shows that fetuses with some forms of CHD have smaller total brain volumes than normal fetuses during the third trimester, a critical period of development during which there is generally an acceleration of brain growth, which requires a lot of energy. Likewise in the Sun L20 study, Magnetic Resonance and Doppler echocardiography studies have confirmed fetal blood flow abnormalities and decreased oxygen delivery, leading to the immaturity of the developing brain. This study evaluated the direct link between decreased brain oxygenation and impaired brain growth in fetuses with CHD, finding an average 32% reduction in brain oxygen consumption in fetuses with CHD (p value<0.001), associated with a 13% mean reduction in brain volume (p value<0.001) and a reduction of the complete standard deviation in the estimated Z score of brain weight (p value<0.001), as well as a lower brain volume in all CHD compared to controls.20 Are duction in venous umbilical oxygen content was evident in fetuses with CHD (p value<0.001), and a failure in oxygen transport from the placenta to the ascending aorta, with a decrease in oxygen saturation fetal brain of 10% (p value<0.001), a mean 6% reduction in umbilical vein oxygen saturation (p=0.0004), which was associated with a mean reductionof17% in delivery of oxygen to the fetal circulation.20 The Masoller N7 study was a case-control study that likewise evaluated the associations between CHD, head biometry, and cerebrovascular blood flow dynamics at the time of CHD diagnosis, in the second trimester of pregnancy.7 In this it was found that a high proportion of fetuses with CHD had a smaller head and still a greater cerebral perfusion in the second trimester of pregnancy. This suggests an early onset of the mechanisms that lead to poorer neurological development later in life. The parameters they evaluated were: biparietal diameter (BPD), head circumference (HC), abdominal circumference (AC) and length of the femur (FL). Cerebral perfusion was evaluated using the middle cerebral artery pulsatility index (MCA-PI), the cerebro placental ratio (CPR) and the fractional moving blood volume (FMBV). These fetuses with CHD showed significantly lower MCA PI and CPRZ scores (−0.23vs.0.34 and−0.37vs.0.30, respectively; p value<0.001) as well as higher FMBVZ score (2.35vs.0.15; p value<0.001). The FMBV>95th percentile was observed in 81.1% of the cases compared to10.5% in controls (p value<0.001), the cases showed a significantly lower Z score of the DBP and HC (P<0.001), in addition a higher proportion of BPD and HC measurements below the 5th percentile compared to controls (51.6% vs. 13.7% and 26.3% vs. 4.2%, respectively; both p value<0.001). This study also found that fetuses with tetralogy of Fallot (TOF) have lower growth rates of the head and estimated fetal weight (EFW), compared to fetuses with HLHS or TGA type CHD. Results similar to those found in the study by Schellen C,21 which identified early changes in the volumes of the fetal total brain (TBV), gray matter (GMV) and subcortical brain (SBV) in fetuses with TOF.21 They also found that the TBV was significantly lower (p value<0.001) in the cases of early TOF (<25 of GA) and late. Additionally, the GMV to SBV ratio decreased in TOF fetuses (p value=0.026) compared to normal fetuses, and ventricular volume (p value=0.0048) as well as external cerebrospinal fluid increased in relation to head size (p value<0.001). They concluded that fetuses with TOF have smaller volumes of gray and white matter, with enlarged spaces of cerebro spinal fluid.
Cohort studies (n=9)
In the analysis of the cohort studies we found results similar to those found in the case-control studies in six of the nine studies. The study by Williams IA22 evaluated the differences in growth between fetuses with and without CHD and the association between growth and early infant ND.21,22 The results of this study demonstrate that fetuses with CHD had lower growth rates compared to controls (see parameters evaluated and evaluation periods in Table 1, this difference being more notable for HC. Fetal biometry variables that were correlated with cognitive score included: AC at 18-26 GA (p value=0.049) and the HC/AC ratio at 18-26 GA (p value=0.02). The language score was significantly correlated with the FL/BPD ratio at 27-33 GA (p value=0.037) and with the HC/AC ratio (p value=0.045). Motor score was not correlated with any biometric variable. These results demonstrated that fetuses with CHD have lower growth rates compared to controls, mainly for HC. Similar results were found in the Masoller N7 study, where they report that fetuses with CHD had DBP and HC in percentiles <5 for gestational age.23 In this study, furthermore, it was shown that fetuses with CHD had significantly lower Z scores for MCA-PI and CPR and a higher Z score for FMBV compared to controls. These same authors previously reported (2015) that fetuses with CHD showed significant changes in brain development, being higher in fetuses with CHD associated with a severe reduction in cerebral oxygenated blood supply, suggesting that this reduction is related to severity of ND deterioration.24 Olshaker H25 reported that fetuses with CHD had smaller cerebellar volumes than healthy fetuses, so they suggested additional research in this group of patients as aradiological marker for long-term out come.25




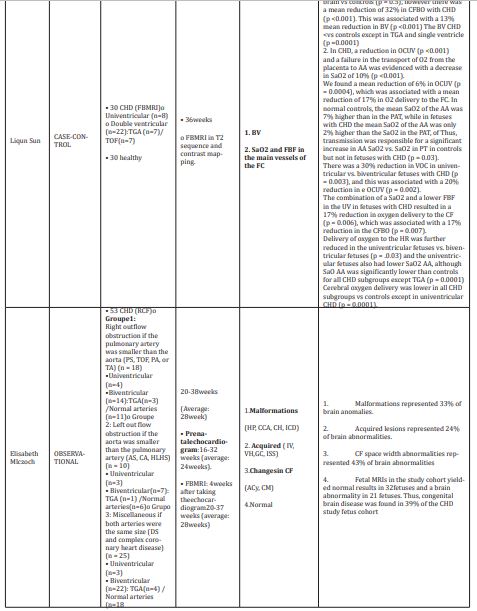
In the study by Ortinau C4 it was demonstrated that fetuses with CHD hadaltered global sulcal patterns in the left hemisphere compared to normal fetuses (p value=0.002).4 This same group in the same year, reported that infants with CHD had a smaller brain volume before being taken to surgery (p value<0.001) in the same way in the MRI at 3 months (p value=0.0001).26 In the previously mentioned studies, it is demonstrated that the alteration of the cerebral structural development in fetuses with CHD can have a prenatal origin, being an important predictor of adverse neuro developmental results. However, the sensitivity and specificity of magnetic resonance imaging of the fetal brain to predict neonatal brain injury is currently unknown. Mlczoch E10 identified the type and incidence of fetal brain pathology in fetuses with prenatal diagnosis of CHD, finding congenital brain disease in 39% of the fetus cohort of the CHD study. However, this study reports limited data on available outcomes.10 Subsequently, M Brossard Racine27 showed that the cerebral anomalies detected in the MRI were significantly more frequent among patients with CHD compared to controls (p value<0.001), and were detected in approximately a quarter (33/144,22.9%) of fetuses with CHD, compared to only 1.5% (3/194) in controls (p value<0.001).27 In 2016, this same research group reported a sensitivity of 27.3%, and a specificity of 88.6%, with positive and negative predictive values of 52.9% and 72.1%,respectively, of conventional fetal MRI, to identify persistent neonatal brain abnormalities, indicating that fetuses with abnormal findings of fetal MRI and CHD images were more likely to have brain abnormalities on neonatal MR images (8 versus 1, p value=0.05).28 These findings suggest that brain abnormalities, detected by MR images in utero, in fetuses with CHD are associated with an increased risk of postnatal preoperative brain injury. However, a substantial proportion of abnormalities on postnatal MR images were not present on fetal MR images.
Clinictrial (n=1)
Hahn E29 investigated the association of fetal growth and cerebrovascular resistance at different periods of gestation, monitoring the ND at 14 months by means of the evaluation with the mental development index (MDI), which assesses cognitive ability and the averages core of the psychomotor development index (PDI), in patients with univentricular heart disease.29 This study pointed to the third trimester as a crucial time for fetal somatic growth and neurological development. It was found that as the gestation progressed the MCA-PI decreased more than would be expected for GA, which shows a decrease in cerebro vascular resistance. In addition, there was a decreased growth in fetal HC later in gestation and MCA-PI was independently and negatively associated with the psychomotor development index. When evaluating the 82 subjects included in the sample, it was found that the DMI scores were significantly lower than the population means for these standardized tests (p value<0.001). No changes were found in the EFW, FL or AC correlated with the ND.
Risk of bias assessment Case-control studies
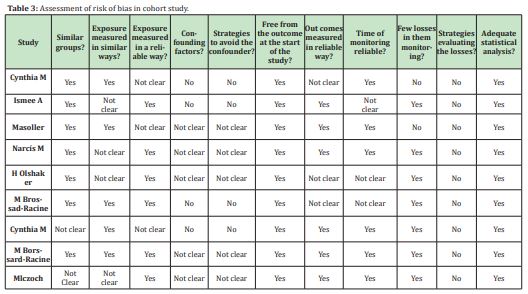
In general, the 6 case-control studies found are intermediate or high risk due to the lack of management of confounding factors and the ways in which the outcome is measured in both cases and controls (Table 2).

Cohort studies
All 9 included cohort studies were at intermediate risk of bias as confounding factors are similarly not managed appropriately (Table 3).
Clinic trials
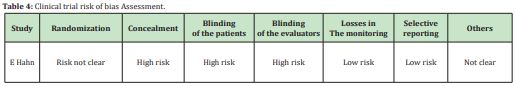
The only included clinical trial29 was at high risk of bias, because the authors do not present methods of randomization or concealment of the evidence used (Table 4).
CHDs represent an important cause of ND alterations in childhood.1-5 Khalil et a described brain disorders diagnosed in fetal neuro imaging for all CHDs, with a prevalence of 34% for TGA and 49% for HLHS.3,9,10 Investigations focused on the alterations in perioperative corrective surgery and cardiopulmonary bypass, however more than half of the children had cognitive and structural deficiencies prior to the surgical procedure, which is why it was raised, with more evidence, that the alterations of the CNS occur since intrauterine life.9,11,12 For all types of CHD a decrease in fetal growth was found, mainly in HC, BPD (Z-score-percentile<5) and CC/AC ratio.22 Morphological findings were associated with decreased cognitive and language development scores in childhood in fetuses between 18-26 weeks of GA. The aforementioned evidences that apart from the oxygenation that CHD presents from the postnatal point of view (cyanotic and non-cyanotic) the morphological alterations lead to a functional alteration that is evidenced in postnatal tests. Growth alterations of the cephalic diameters mentioned would evident when noxa occurs early in gestation as a result of CHD.29,7,23,24 It is important to highlight that our findings are complemented by the results found in the literature where up to 59% of newborns with TGA and HLHS (Figure 1), have a delay in brain growth in the third trimester.3 It is striking how Williams22 although they report alterations in fetal biometric measurements, they do not show a direct association with an alteration in ND in childhood.22,29,23 The only clinical trial published in this regard,29 showed a postnatal alteration, in the cognitive capacity and psychomotor development of newborns with univentricular CHD after follow-up for 14 months.29 Patients with ventricular hypoplasia will have cognitive impairment during postnatal monitoring. Several authors have evaluated brain and cerebrospinal fluid volumes as an alteration of the CNS secondary to a decrease in cerebral perfusion.6,21 Decreased brain volumes have been shown in fetuses affected with CHD, much more evident in fetuses with HLHS and TOF, in which an alteration in brain volumetry predominantly seen in TBV is found, ICV, white-matter-volume, and cerebellar-volume.6,13,21 They do not report significant differences in the volumes of cortical/subcortical or ventricular gray matter.21,23-25 The above findings are striking since the measurements of brain volumes are reproducible measurements on MRI by specialists in reading fetal imaging studies. Donofrio11 established the difference between brain abnormalities due to hypoxic-ischemic lesions vs. acquired-malformations.11 Severe structural injuries, such as holoprosencephaly, were not excluded resulting in an overvaluation of findings, with an association of 39% congenital brain disease in fetuses with CHD.10 By not excluding CNS pathology, it is very difficult to determine if the origin of the lesion could have been in CHD, therefore, it is evident how previous studies suggest that cerebral morphological evaluation equires to determine which lesions are secondary to CHD oxygenation disorders.
Ortinau4 conducted a study in which they compared cortical development by describing the normal surcal pattern with the finding that fetuses with CHD had altered surcal patterns predominantly in the left hemisphere (p value=0.002). These differences are represented by alteration from the early formation of the grooves and in the position of their precursors (p value=0.006) as well as the inter surcal relationships (p=0.033).4 In our review, it was found three studies that evaluated the surcal pattern with similar findings. The greatest involvement remains persistent in fetuses with univentricular CHD, where the cingulate sulcus is not visible at the 28 weeks of GA and the superior frontal sulcus is not visible at the 30 weeks, opposite in healthy fetuses.13 Furrow patterns were predominantly altered in the left hemisphere with significantly decreased depths for parieto-occipital, cingulate, and calcarine fissures.4,23,24
The most studied parameter of cerebral perfusion by means of Doppler is the alteration of the ACM-IP and the CPR. This alteration appears in fetuses with HLHS, AS or AAH, represented as a decrease in PI beyond that expected for GA.29 The above implies a compensatory mechanism of cerebral vasodilation as a consequence of inadequate perfusion due to an alteration in cardiac structurality, the alteration in univentricular vs. biventricular CHDs being more representative and being an independent factor of abnormal brain development.7,23,24 This finding, although it seems highly related to central hypo perfusion secondary to CHD, appears frequently in fetuses with IUGR who present al eft ventricular dysfunction that alters the PSV-MCA without presenting an alteration in blood viscosity. This hemodynamic finding could be increased in fetuses with both abnormalities CHD/IUGR. If the fetus with CHD also presents IUGR, its prenatal neurological prognosis would be deleterious in the ND.3,4 Alterations such as periventricular leukomalacia, cerebralatrophy and white-matter damage, evidenced both prenatally and postnatally, were findings in which most of the authors agree (Figure 1).3 Regarding the evaluation and comparison of prenatal MRI studies compared to postnatal findings, one study evaluated prenatal vs. postnatal performance with a finding of sensitivity 27.3%, specificity 88.6%, PPV52.9%, and NPV of 72.1% to detect brain abnormalities priortobirth.28 Likewise, the values for prenatal NS in terms of sensitivity, specificity, NPV and PPV are 96%, 87%, 93% and 93% respectively.30,31 Not all disorders are identified prenatally, which could be a consequence of technical/observer limitations.
This systematic review has brought together the most relevant studies regarding the important effect of hypoxia in utero secondary to CHD evident in prenatal examinations. Our findings suggest that in the included literature it is evident how the lesions occur in a prenatal stage and are corroborated at birth, demonstrating how cardiac surgery was not the cause of said lesions but CHD per se, however cardiac surgery could present alterations in the cerebral circulation with consequences. Fetuses with isolated CHD present structural neurological alterations since intrauterine life, even from early stages of fetal development, evidenced by NS and fetal-MRI. This highlights the importance of studying the CNS of fetuses with CHD and the relationship between structural brain alterations and ND impairment since prenatal stages. Long-term prospective multicenter cohort studies are required, with prenatal and postnatal evaluation that allows evaluating the impact of these structural alterations on the incidence of cognitive development disorders.
None.
None.
Authors declare that there is no conflict of interest.
- 1. Fenna AR Jansen, Sheila MP Everwijn, Robert Scheepjens, et al. Fetal brain imaging in isolated congenital heart defects - a systematic review and meta-analysis. Prenat Diagn. 2016;36(7):601–613.
- 2. Hoffman JIE, Kaplan S. The incidence of congenital heart disease. J Am Coll Cardiol. 2002;39(12):1890–1900.
- 3. Khalil A, Suff N, Thilaganathan B, et al. Brain abnormalities and neuro developmental delay in congenital heart disease: Systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2014;43(1):14–24.
- 4. Ortinau C, Rollins C, Gholipour A, et al. Early Emerging Sulcal Patterns are Atypical in Fetuses with Congenital Heart Disease. Cereb Cortex. 2018;1–12.
- 5. Mebius MJ, Kooi EMW, Bilardo CM, et al. Brain injury and neuro developmental outcome in congenital heart disease: A systematic review. Pediatrics. 2017;140(1): e20164055.
- 6. Limperopoulos C, Tworetzky W, McElhinney DB, et al. Brain volume and metabolism in fetuses with congenital heart disease: Evaluation with quantitative magnetic resonance imaging and spectroscopy. Circulation. 2010;121(1):26–33.
- 7. Masoller N, Martínez JM, Gómez O, et al. Evidence of second trimester changes inhead biometry and brain perfusion in fetuses with congenital heart disease. Ultrasound Obstet Gynecol. 2014;44(2):182–187.
- 8. Martinez Biarge M, Jowett VC, Cowan FM, et al. Neuro developmental outcome in children with congenital heart disease. Semin Fetal Neonatal Med. 2013;18(5):279–285.
- 9. KhalilA, Bennet S, Thilaganathan B, et al. Prevalence of prenatal brain abnormalities in fetuses with congenital heart disease: a systematic review. Ultrasound in obstetrics & gynecology. 2016;48(3):296–307.
- 10. Mlczoch E, Brugger P, Ulm B, et al. Structural congenital brain disease in congenital heart disease: Results from a fetal MRI program. Eur J Paediatr Neurol. 2013;17(2):153–160.
- 11. Donofrio MT, Massaro AN. Impact of Congenital Heart Disease on Brain Development and Neurodevelopmental Outcome. Int J Pediatr. 2010;2010:1–13.
- 12. Miller SP, Mc Quillen PS, Hamrick S, et al. Abnormal brain development in newborns with congenital heart disease. N Engl J Med. 2007;357(19):1928–1938.
- 13. Clouchoux C, Du Plessis AJ, Bouyssi Kobar M, et al. Delayed cortical development in fetuses with complex congenital heart disease. Cereb Cortex. 2013;23(12):2932–2943.
- 14. Beca J, Gunn J, Coleman L, et al. Pre-Operative Brain Injury in Newborn Infants With Transposition of the Great Arteries Occurs at Rates Similar to Other Complex Congenital Heart Disease and Is Not Related to Balloon Atrial Septostomy. J Am Coll Cardiol. 2009;53(19):1807–1811.
- 15. Limperopoulos C, Majnemer A, Shevell MI, et al. Neuro developmental status of newborns and infants with congenital heart defects before and after open heart surgery. J Pediatr. 2000;137(5):638–645.
- 16. Miller S, Mc Quillen PS, Hamrick S, et al. Abnormal Brain Development in Newborns With Congenital Heart Disease. Surv Anesthesiol. 2008;52(3):127–128.
- 17. Luis Salvador Carulla, Geoffrey M Reed, Leila M Vaez Azizi, et al. Intellectual developmental disorders: towards a new name, definition and framework for “mental retardation/intellectual disability” in ICD-11. World Psychiatry. 2011;10(3):175–180.
- 18. Lunsky Y, De Oliveira C, Wilton A, et al. High health care costs among adults with intellectual and developmental disabilities: a population-based study. J Intellect Disabil Res. 2019;63(2):124–137.
- 19. Weissmann-Brenner A, Mitlin A, Hoffman C, et al. Assessment of the association between congenital heart defects and brain injury in fetuses through magnetic resonance imaging. Isr Med Assoc J. 2020;22(1):27-31.
- 20. Sun L, Macgowan CK, Sled JG, et al. Reduced fetal cerebral oxygen consumption is associated with smaller brain size in fetuses with congenital heart disease. Circulation. 2015;131(15):1313–1323.
- 21. Schellen C, Ernst S, Gruber GM, et al. Fetal MRI detect early alterations of brain developmentin Tetralogy of Fallot. Am J ObstetGynecol. 2015;213(3):392e1–392e7.
- 22. Williams IA, Fifer WP, Andrews HF, et al. Growth and Neurodevelopmental Outcome in Congenital Heart Disease. Pediatr Cardiol. 2015;36(6):1135–1144.
- 23. Masoller N, Sanz Cortés M, Crispi F, et al. Mid-gestation brain Doppler and head biometry in fetuses with congenital heart disease predict abnormal brain development at birth. Ultrasound Obstet Gynecol. 2016;47(1):65–73.
- 24. Masoller N, Sanz Cortés M, Crispi F, et al. Severity of Fetal Brain Abnormalities in Congenital Heart Disease in Relation to the Main Expected Pattern of in utero Brain Blood Supply. Fetal Diagn Ther. 2016;39(4):269–278.
- 25. Olshaker H, Ber R, Hoffman D, et al. Volumetric brain MRI study in fetuses with congenital heart disease. Am J Neuroradiol. 2018;39(6):1164–1169.
- 26. Ortinau CM, Mangin Heimos K, Moen J, et al. Prenatal to postnatal trajectory of brain growth in complex congenital heart disease. Neuro Image Clin. 2018;20:913–922.
- 27. M Brossard Racine, AJ du Plessis, G Vezina. Prevalence and spectrum of in utero structural brain abnormalities in fetuses with complex congenital heart disease. Am J Neuroradiol. 2014;35(8):1593–1599.
- 28. M Brossard Racine, AJ du Plessis, G Vezin. Brain Injury in Neonates with Complex Congenital Heart Disease: What Is the Predictive Value of MRI in the Fetal Period? M. AJNR Am J Neuroradiol. 2016;176(5):1338–1346.
- 29. Hahn E, Szwast A, Cnota J, et al. Association between fetal growth, cerebral blood flow and neuro developmental outcome in univentricular fetuses. Ultrasound Obstet Gynecol. 2016;47(4):460–465.
- 30. Mari G, Picconi J. Doppler Vascular Changes in Intrauterine Growth Restriction. Semin Perinatol. 2008;32(3):182–189.
- 31. Malinger G, Ben Sira L, Lev D, et al. Fetal brain imaging: A comparison between magnetic resonance imaging and dedicated neurosonography. Ultrasound Obstet Gynecol. 2004;23(4):333–340.
exp White Matter/White Matter.tw (White adj3 Matter).tw
(occipitofrontal circumference).tw (cephalic circumference).tw
Exp Brain Diseases/Brain.tw (Intracranial adj3 Disorders).tw (Intracranial Disorder).tw (Encephal* Diseases).tw (Encephal* adj3 Diseases).tw Encephal*.tw
exp Central Nervous System/"Central NervousSystem".tw Cerebrospinal Axi.tw (Cerebrospinal adj3 Axi).tw exp Brain/ exp Magnetic Resonance Spectroscopy/Magnetic Resonance.tw (Magnetic adj3 Resonance).tw MRSpectroscopy.tw (MR adj3 Spectroscop*).tw (NMR adj3 Spectroscop*).tw NMR Spectroscop*.tw
exp Heart Diseases/(Heart Disease).tw (Heart adj3 Disease).tw (Cardiac Disease).tw
Cardiac adj3 Disease).twcardiopathy.tw exp Fetus/Fetus.twFetal.tw

